By Rodolfo Roman-Guzman (February 18, 2026)

Late last year, I had the privilege of caring for an extraordinary couple. She, 74 years old, was a professional flutist for much of her life and now lives with a severe amnestic syndrome secondary to advanced Alzheimer’s disease. He is a visual artist. Together, they share a deeply captivating bond, and it is entirely to their openness and generosity in sharing their story that I owe this essay—and the learning that accompanies it.
Unable to dress herself, to shower independently, or to use the bathroom appropriately—and frequently experiencing episodes of disorientation even within her own home—she nevertheless remains “happy and joyful.” There is in her a profound amnestic state; yet when she takes her transverse flute and places it in her hands, something changes. She begins to play, in a scene that seems to lift her out of the neurodegenerative process, into a space where music becomes the sole bearer of truth. In that reality, not only do her hands, mind, and mouth coordinate with remarkable precision, but the music becomes embodied in her, projecting who she is, who she has always been, and how she will be remembered: music.
This was not merely something described during a clinic visit. A few days later, he graciously sent me a series of videos in which she can be seen playing the flute. One of them, in particular, evoked the profound meaning of love within a partnership: he seated at the piano, she with her flute, sharing an activity that has become part of their daily rhythm. They play together, and for her, playing the flute is one of the few activities she can still perform independently—while at the same time fully sharing it with her husband.
This activity, however, is not merely an exercise in repetition. When she plays the flute, she is able to improvise—with a degree of judgment and autonomy that is no longer possible for her in other areas of daily life. In one of the videos, she can even be seen improvising “Happy Birthday to You.” There are pauses, mistakes—natural, as we all have in other domains of life—but she retains mastery of the instrument, continues playing, and smiles. Music reaffirms her identity and, in those moments, confirms her place as an individual.
Music, as a defining element of the individual, thus becomes the central theme of the connections I will attempt to unfold here.
A few days later, as I was walking home on a snowy day during my first winter in Canada, I found myself overtaken by an introspective—perhaps even nostalgic—reflection. I thought of my flutist patient and, at the same time, recalled a scene from The Pianist, directed by Roman Polanski. It is the moment when Szpilman is hiding in a room inside an apartment where there is a piano he is strictly forbidden to touch, since even the slightest sound could betray him. The neighbours might report the presence of an intruder, and if discovered to be Jewish, the Germans would annihilate him without hesitation.
This scene is sublime because, by that point in the story, Szpilman—like most members of his community—has already lost everything. First and foremost, he has lost his family: his father, his mother, his siblings. He has lost his home, his material possessions, his work, and his social standing. Yet along that harrowing journey of loss, hunger, and despair, there is something he has not lost: music.
Szpilman sits before the piano, opens the keyboard, and—in a kind of hallucinatory reverie—begins to play in the air. He does not press a single key, yet he hears every note with absolute clarity in the ear of his mind: Andante spianato et Grande polonaise brillante, Op. 22, by Chopin. In that trance-like moment, however, he is not alone. He interacts with other musicians who inhabit the echoes of his memory and reappear in an imagined concert that offers him—if only for an instant—a creative refuge from his inner torment.
The story of The Pianist is rooted in the lived reality of the Holocaust. Władysław Szpilman, a composer and pianist only 35 years old at the time, wrote his memoir in 1946 under the original title Death of a City as a form of personal catharsis in the aftermath of the psychological trauma inflicted by the Nazi occupation of Poland—an occupation that led to the death of more than half a million Jews in Warsaw, including his own family.
Shortly thereafter, however, the book was restricted by the Stalinist authorities in Poland and remained in obscurity for decades. It was not until the late 1990s that his son, Andrzej Szpilman—convinced that his father’s work deserved to see the light again—promoted its translation into English under the title The Pianist: The Extraordinary Story of One Man’s Survival in Warsaw, 1939–1945.
In 2002, with a screenplay adapted by Ronald Harwood and directed by Roman Polanski—who himself survived the Nazi occupation of Kraków as a child—the story reached the screen. The Pianist won the Academy Awards for Best Director, Best Adapted Screenplay, and Best Actor. Adrien Brody’s extraordinary performance ultimately solidified a cinematic work that, rather than merely recreating a historical episode, embodies an intimate experience of survival.
The Pianist offers us, from another perspective, a reflection on music as a creative force when everything else appears lost. Music possesses an intrinsic narrative capacity, and it is no coincidence that the work of Chopin—often regarded as “the poet of the piano”—accompanies Szpilman throughout his story of survival.
Toward the end of the film, after not only the Jewish insurgents of the ghetto have fallen but also the Polish resistance fighters who at one point joined the uprising, Szpilman is forced to flee once again and, paradoxically, finds refuge within the ruins of the ghetto itself. Malnourished, dehydrated, exhausted, in shock and terrified, he shelters in a destroyed building where he is discovered by Captain Wilm Hosenfeld, an officer of the German army.
Standing before him—perhaps imagining this to be the end—Szpilman is interrogated: What are you doing here? Who are you? What do you do? He replies, “I am a pianist.” Hosenfeld leads him into a room with a piano and asks him to play. Szpilman sits down and, after nearly two years without having played a single piece, begins to perform Chopin’s Nocturne in C-sharp minor. In the film, however, Polanski adjusted the repertoire, and Adrien Brody performs Ballade No. 1 in G minor, Op. 23. I will not dwell here on the cinematic reasons for that change.
Whatever the piece, the scene is sublime. A man who has lost everything, facing someone who could be his executioner and knowing that this may be the final moment of his existence, sits at the piano and does what he knows best—what defines him as an individual: he plays. And in playing, he becomes once again what he is—a pianist.
This scene of courage and human expressiveness in the face of adversity bears, without doubt, a profound connection to my patient and her flute. In both cases, identity seems to return the moment they touch their instrument.
For Szpilman, the assault was not biological but psychological: the violence of the Holocaust functioned as an extreme trauma—an archetype of human suffering. In my patient—as in so many individuals living with Alzheimer’s disease—it is neurodegeneration that progressively erodes episodic memory.
In both, however, it is music that keeps identity afloat.
And the preservation of musical memory is neither an isolated anecdote nor an exceptional romantic phenomenon. It has been described in various neurological conditions, with one of the most emblematic cases being that of Clive Wearing. In Musicophilia (2007), Oliver Sacks recounts the story of this English musician who, at the age of 47—just as he was reaching the height of his career—contracted herpetic encephalitis that bilaterally destroyed the medial regions of his temporal lobes, including both hippocampi: structures essential for the consolidation of new memories. Since then, Wearing has suffered from one of the most severe amnesias ever documented. He cannot form new memories and has lost much of his past. He lives in a fragmented present, where each moment is experienced as if it were the first, often with profound anguish.
Yet—and herein lies the key—his musical capacity remains extraordinarily intact.
Wearing can sit at the piano and fluently perform complex works. He can read musical scores, sing in tune, improvise, and even conduct a choir. He has not lost the musical skills acquired before his illness, and he is even capable of refining his performance through practice, despite retaining no conscious memory of having practiced. Oliver Sacks notes that if he is asked minutes later what piece he has just played, Wearing may even deny having played anything at all; he has lost the ability to verbalize memories, to declare facts.
Music, however, manages to reorganize him. With his hands on the piano, Wearing ceases to be a man trapped in inescapable oblivion. In that moment, continuity reemerges. Musical performance activates auditory–motor, procedural, and emotional networks that do not depend exclusively on the damaged hippocampus. In other words, although his episodic memory is devastated, his procedural memory and his musical representations distributed across cortical and subcortical networks remain functional.
But so that we may reflect on and assimilate this information together, I would like to explain—supported by the following diagrams and images—a bit more deeply the neuroanatomical and functional substrate underlying these phenomena.
Memory systems can be divided, at a first level, into short-term memory and long-term memory.
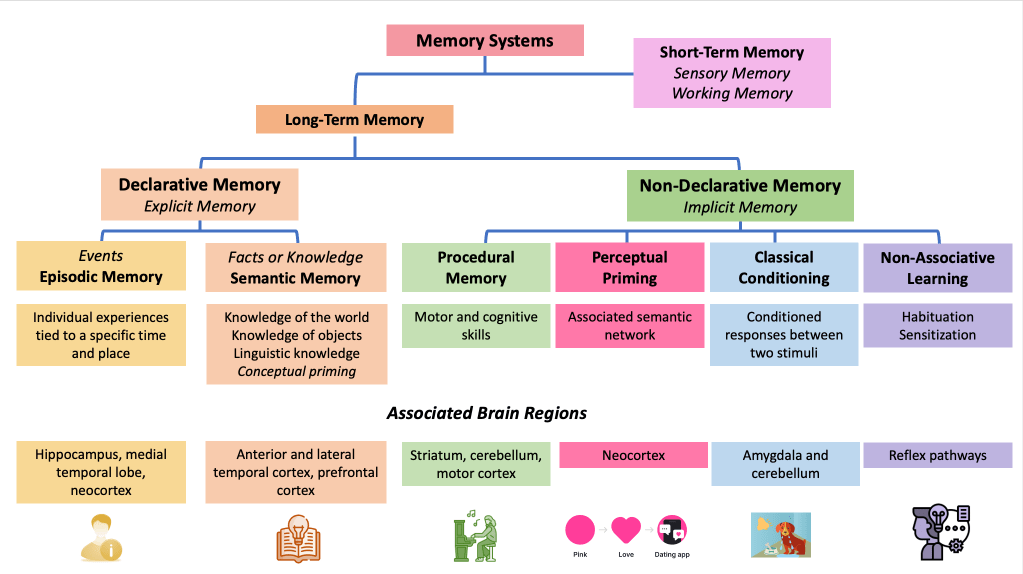
Image credit: Original image, based on Alessandro L, Ricciardi M, Chaves H, Allegri RF. Acute amnestic syndromes.Journal of the Neurological Sciences. 2020;413:116781. https://doi.org/10.1016/j.jns.2020.116781
Short-term memory²—also referred to as working memory—allows us to temporarily retain information while performing a specific task. However, working memory is not merely a transient storage system; rather, it is a dynamic, multicomponent architecture that enables us to actively maintain and manipulate what we perceive in the present moment (see image below).
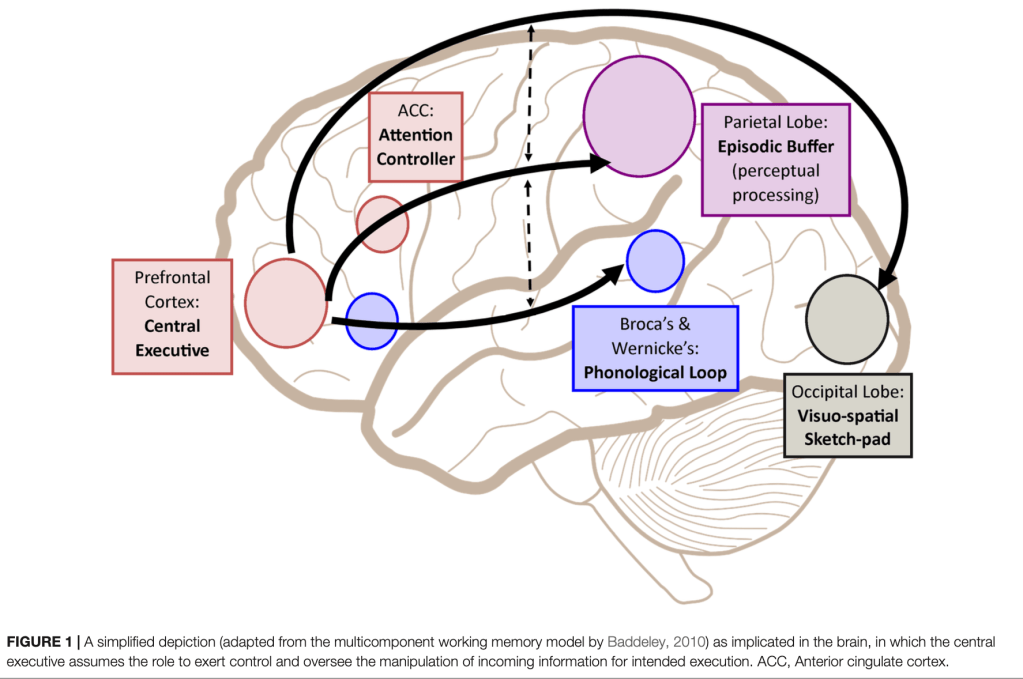
Image credit: Chai WJ, Abd Hamid AI, Abdullah JM. Working Memory From the Psychological and Neurosciences Perspectives: A Review. Frontiers in Psychology. 2018;9:401. https://doi.org/10.3389/fpsyg.2018.00401
When we hear a sequence of words, read a musical score, or attempt to solve a problem, different brain regions coordinate to keep that information accessible while we transform it. The phonological loop sustains verbal material through frontotemporal networks; the visuospatial sketchpad maintains spatial and visual representations via parieto-occipital circuits; and the central executive, primarily located in the prefrontal cortex, supervises, selects, and inhibits distractors, modulating cognitive effort in conjunction with the anterior cingulate cortex, which serves as an attentional control hub.
Meanwhile, the episodic buffer operates as a temporary integrative workspace in which these subsystems converge and connect with information previously stored in long-term memory. Far from functioning as isolated compartments, working memory and long-term memory are in constant interplay: what we manipulate in the present depends on networks consolidated in the past, and what we actively rehearse today may become tomorrow’s enduring memory.
On the other hand, long-term memory³ stores information that belongs to the past. It can be subdivided into two major systems.
Declarative, or explicit, memory refers to the type of memory we can express in words. It is the memory we are able to recount, describe, and verbally communicate. Within this system, we distinguish:
Episodic memory, which corresponds to our autobiographical experiences situated in a specific time and place and depends on structures of the medial temporal lobe, particularly the hippocampus.
Semantic memory, which represents our knowledge of the world—concepts, meanings, facts, words, and information—and relies on the anterior and lateral temporal cortex, with additional involvement of the prefrontal cortex.
The following images—drawn from the classic article “Memory Dysfunction” by Budson and Price, published in The New England Journal of Medicine (2005)—serve to visually illustrate the principal neuroanatomical structures involved in the different memory systems.
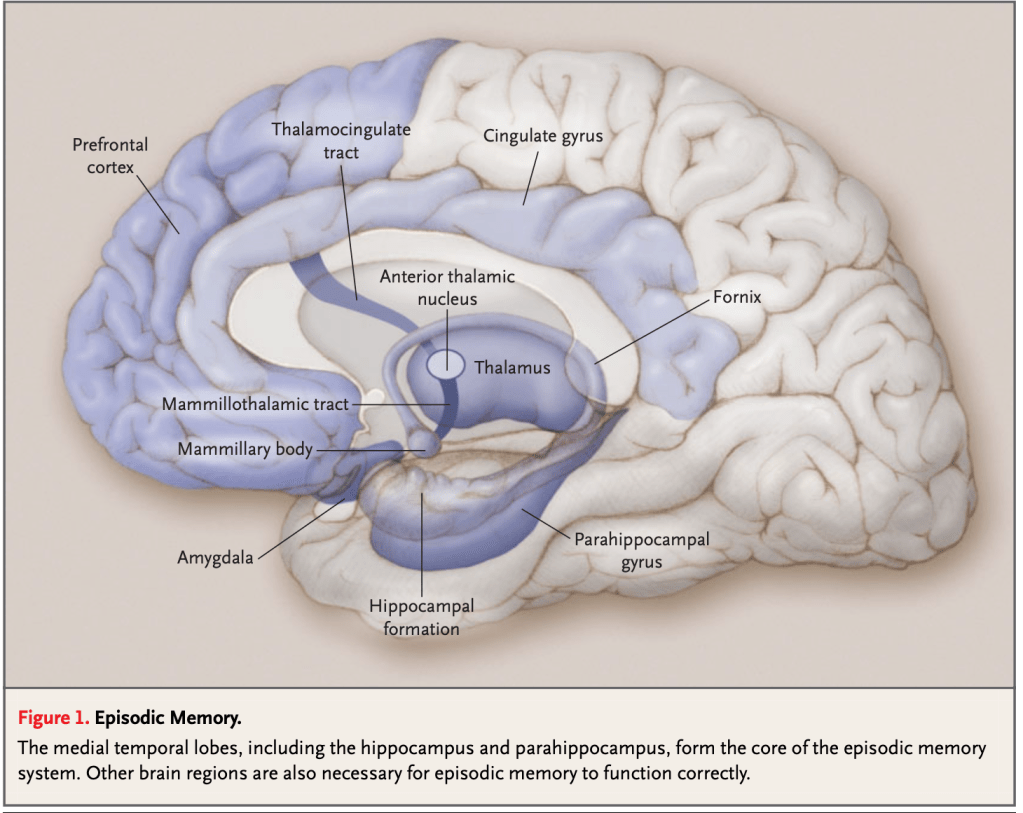
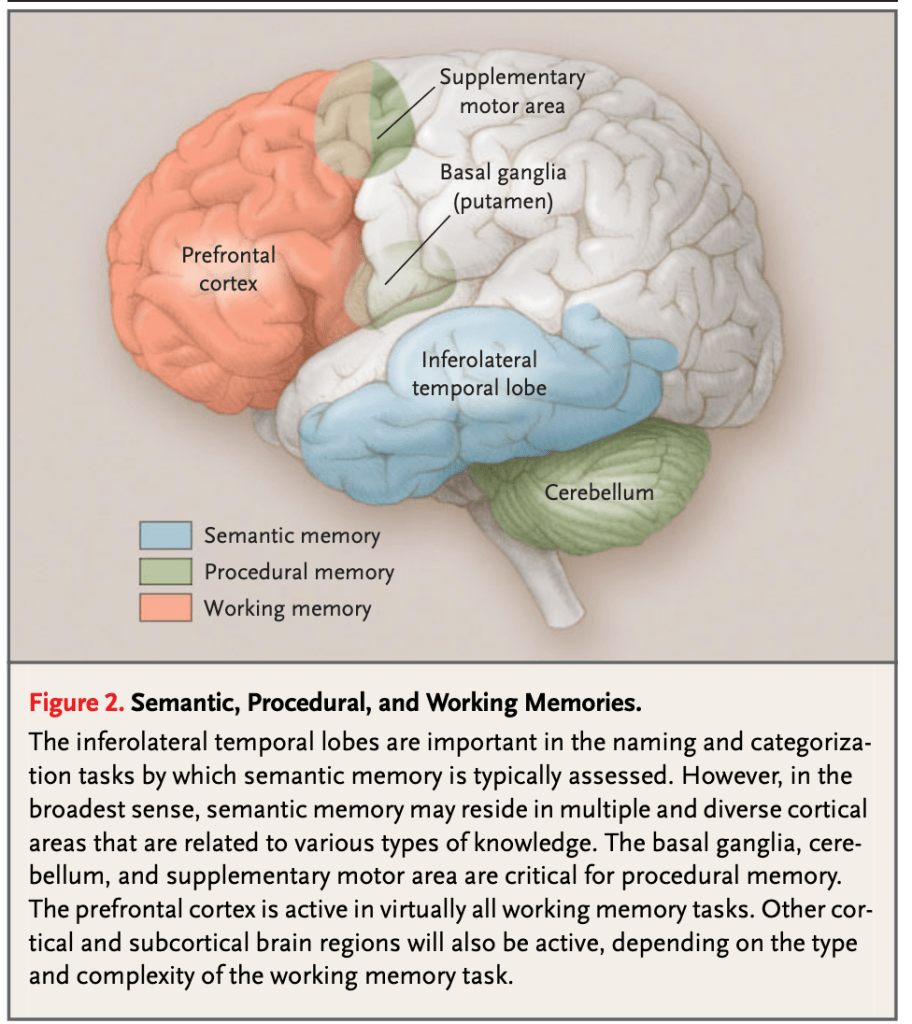
The second major system is nondeclarative or implicit memory, which does not require conscious recollection. Unlike declarative memory, it does not depend on the ability to verbalize what has been learned or to recount a past experience. Rather, it is a system that sustains skills and forms of learning expressed through performance rather than narrated recall. And it is precisely this system that explains why, in my patient and in Clive Wearing, music remains alive despite their amnestic syndrome.
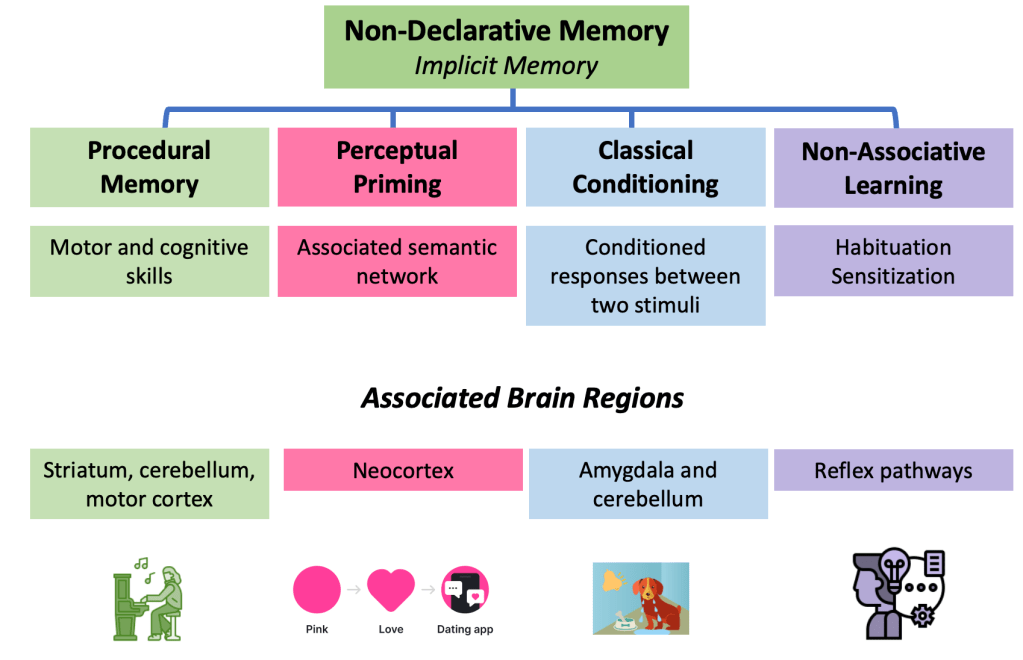
Procedural memory encompasses the complex motor and cognitive programs that allow us to learn how to ride a bicycle, master a tool, type on a keyboard, or skillfully perform a musical piece on the transverse flute. Its anatomical substrate is not primarily located in the hippocampus or the medial temporal cortex, but rather in a distributed network that includes the striatum, the cerebellum, and the motor cortex, among other structures.
It is precisely this distributed organization that explains why a patient with severe impairment of episodic memory may nonetheless retain the ability to perform a skill learned decades earlier.
We owe these concepts to the study of the most emblematic case in the history of cognitive neurology: Henry Molaison⁴—known for decades as H.M.—who in 1953 underwent an experimental surgical procedure to treat his refractory epilepsy. At that time, diagnostic tools were limited; magnetic resonance imaging did not yet exist, and intracranial electrophysiology was not part of the systematic clinical approach.
Based on prior clinical observations and on the hypothesis that the medial temporal lobes played a key role in the generation of epileptic seizures, neurosurgeon William Beecher Scoville decided to perform an extensive bilateral resection of the medial temporal structures. The procedure included large portions of the hippocampus, the amygdala, and the entorhinal cortex.
The intervention succeeded in significantly reducing the frequency of his epileptic seizures. However, it produced an unexpected and devastating consequence: severe anterograde amnesia. After the surgery, Molaison was unable to form new declarative memories—both episodic and semantic.
Paradoxically, his case revealed something crucial for science. Although he could not remember having learned a task, he was nonetheless able to improve at it with practice. This phenomenon was carefully documented by neuropsychologist Brenda Milner through the now-iconic mirror-tracing task. In this experiment, H.M. was asked to trace the outline of a five-pointed star with a pencil while viewing only the reflection of his hand in a mirror, forcing him to invert his usual visuomotor coordination (see illustrative image below).
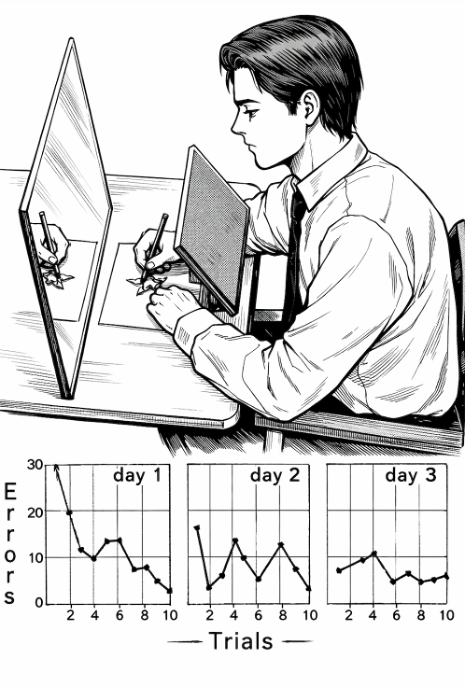
At first, he made numerous errors; his movements were clumsy and disorganized. Yet across repeated trials, his performance improved progressively and consistently, and this improvement was sustained over consecutive days. Most striking of all, each time he returned to the laboratory, H.M. insisted that he had never performed the task before. He had no conscious recollection of the prior experience—yet his brain had learned.
It is thanks to H.M.—and to the pioneering work of Dr. Brenda Milner in 1962—that we now understand memory not as a single, unified entity, but as a constellation of distinct systems. H.M.’s case demonstrated that the structures of the medial temporal lobe are essential for declarative memory. In contrast, the learning of skills—such as tracing a figure in a mirror or performing a musical piece—depends on distributed circuits that include the basal ganglia, the cerebellum, and the motor cortex.
This distinction fundamentally transformed the neuroscience of memory: what we can narrate does not always coincide with what we know how to do. It is one thing to remember an experience; it is quite another to embody a skill.
Allow me here to share a personal anecdote. My paternal grandfather passed away one November at the age of 74. The summer before his death, we took a family trip to the beach. I was a child at the time. I remember his slow gait; he showed no evident cognitive decline, but his motor abilities were no longer what they once had been.
At the pool, my father invited him to swim. What happened next surprised me deeply: that man who walked with such difficulty began to move through the water with remarkable fluidity, without apparent effort.
“What is well learned is never forgotten,” he said. “I used to swim a great deal in the river when I was a boy like you.”
And perhaps he was right—at least with respect to procedural memory. For a few minutes, that frail older man seemed truly at ease in the water. It was not an act of conscious recollection; he was not “remembering” how to swim in the declarative sense of the term. Rather, it was motor learning unfolding naturally, sustained by circuits involving the motor cortex, the cerebellum, and the basal ganglia. The skill remained intact, even as the body bore the marks of time.
But let us return to music. The interaction between music and the brain is far more complex than the mere acquisition of a motor task. The mental machinery that allows us to understand music—to anticipate a chord, to feel the tension of a dissonance, to synchronize the body with a pulse—engages distributed networks that integrate auditory perception, temporal prediction, emotion, and reward⁵. It is no surprise that this complexity was recognized as an enigma even by Charles Darwin. In The Descent of Man, he wrote:
“Since neither the enjoyment nor the capacity to produce musical notes are faculties of the least direct utility to man in relation to the ordinary habits of his life, they must be ranked among the most mysterious with which he is endowed.”
For Darwin, music did not appear to fulfill any obvious practical function; it was a mysterious faculty. Yet years later, in his autobiography, he reflected with a certain melancholy on his own loss of aesthetic sensitivity:
“…if I had to live my life again, I would make it a rule to read some poetry and listen to some music at least once every week; for perhaps the parts of my brain now atrophied would thus have been kept active through use. The loss of these tastes is a loss of happiness, and may possibly be injurious to the intellect, and more probably to the moral character, by enfeebling the emotional part of our nature.”
It is difficult not to recognize the acuity of this intuition. Without access to neuroimaging tools or models of neural networks, Darwin suspected that disuse might affect brain regions linked to emotion and aesthetic pleasure. Today we know that musical experience recruits not only the auditory cortex, but also frontal predictive circuits and mesolimbic dopaminergic systems associated with reward. Music is not a peripheral cognitive luxury; it is an experience that synchronizes perceptual, motor, and emotional networks within an extraordinarily sophisticated dynamic framework.
Music reaches the brain through the ears, which transform vibratory air waves into electrical signals that ascend along the auditory pathway to the primary auditory cortex, located in Heschl’s transverse gyrus. Up to this point, the process does not differ from that of any other sound. But then a fundamental question arises: how does our brain distinguish music from other acoustic stimuli?
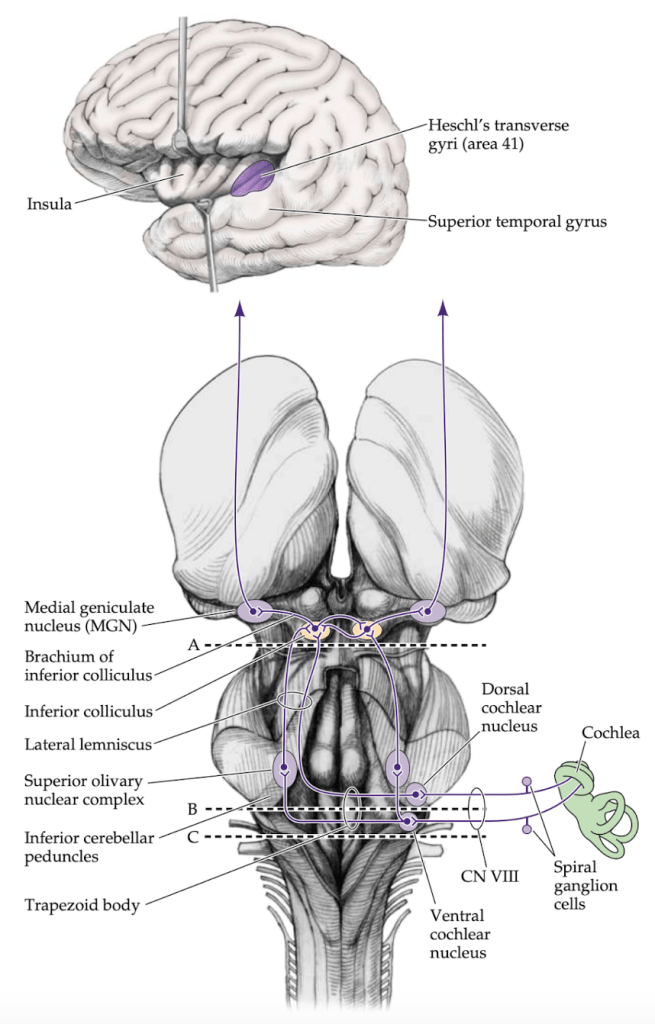
Image credit: Blumenfeld H. Neuroanatomy Through Clinical Cases. 3rd ed. Yale University School of Medicine.
One might say, in a simplified way, that music is nothing more than intentionally organized sound. Yet the brain is not a passive spectator that “computes” music as if solving an equation. It is a dynamic system that resonates with it.
When we listen to music, a single “musical center” does not become active; rather, a distributed network is engaged, including the auditory cortex, frontal regions, motor areas, the basal ganglia, the cerebellum, and structures of the limbic system. These networks form functional loops between the temporal and frontal cortices, enabling the integration of what is heard with what is expected to be heard—or with what we remember having heard before.
Working memory plays a crucial role here. Unlike vision—where objects can remain stationary before us—sound is ephemeral: it vanishes the moment it is produced. For a musical sequence to acquire meaning, the brain must actively maintain previous sounds while processing new ones.
Studies in non-human primates show that monkeys possess a limited capacity to retain auditory events in working memory, especially when compared to their visual memory. Humans, by contrast, demonstrate a remarkable ability to sustain auditory information and relate a present sound to one that occurred seconds—or even minutes—earlier. This difference may help explain why our species developed complex combinatorial auditory systems such as language and music.
From a structural standpoint, music can be analyzed into three major elements: melody, harmony, and rhythm⁶. Melody organizes pitch into sequences with contour and direction; harmony establishes simultaneous relationships between sounds, generating stability or tension; and rhythm structures time through patterns of duration and accentuation. These components do not operate in isolation; rather, they interact across multiple hierarchical levels to produce a unified musical experience—both cognitive and emotional.
Yet this organization is not merely abstract; it rests upon a concrete neural architecture.
Pitch perception begins in the primary auditory cortex, within Heschl’s transverse gyrus, where a precise tonotopic organization exists: different frequencies activate specific cortical regions. However, musical pitch perception extends beyond simple frequency detection. The identification of melodic contour—whether a note rises or falls relative to another—engages secondary auditory regions in the superior temporal gyrus and planum temporale, with greater involvement of the right hemisphere in tasks requiring fine-grained pitch discrimination.
Through connections to inferior frontal regions—particularly within the frontal lobe—the brain not only encodes the present frequency but actively compares the current sound with the one just heard. Here, auditory working memory emerges: maintaining one note in mind while processing the next is essential for melody to exist. Without that transient retention, there would be only isolated sounds.
Harmony, in turn, requires an additional level of integration. Processing chords or simultaneous tonal relationships depends on the interaction between bilateral auditory areas and frontal regions involved in structural analysis. When a chord generates tension or resolution, that evaluation is not purely acoustic; it engages frontotemporal circuits that detect culturally learned regularities and compare the present stimulus with internalized patterns.
Rhythm engages yet another dimension of the musical brain. Although it also begins in the auditory cortex, it quickly recruits the supplementary motor areas, the basal ganglia, and the cerebellum. Even when we remain physically still, these motor regions become active during rhythmic listening. The brain does not merely hear the pulse—it simulates it bodily.
This audio–motor interaction allows us to internalize musical time and anticipate the next accent. The cerebellum contributes to fine temporal calibration, while the basal ganglia participate in pulse detection and metric regularity.
Thus, from the very moment of perception, music is already a distributed experience—temporal, frontal, motor, and limbic. There is no single “musical center,” but rather networks in constant dialogue through specific connective pathways: fascicles linking temporal and frontal cortices, subcortical circuits synchronizing time and action, and limbic projections assigning emotional value.
And here something fundamental occurs for understanding musical memory: these very perceptual circuits are the ones that allow music to leave a trace. Memory does not arise after perception; it emerges from it.
Each time a melodic sequence is maintained in working memory, each time a rhythmic pattern is correctly anticipated, the synaptic connections sustaining that prediction are reinforced. Repetition stabilizes frontotemporal circuits; practice consolidates audio–motor networks; emotion strengthens encoding through limbic systems.
In other words, the cerebral architecture that perceives music is the same architecture that, over time, remembers it.
If this is already fascinating at the perceptual level, it becomes even more complex when we turn to those who not only listen to music but produce it⁷. Playing an instrument entails a constant interaction between auditory and motor systems, engaged in a bidirectional dialogue of feedback and anticipation.
In trained musicians, this interaction becomes even more tightly coupled. Listening to a well-learned piece activates not only the auditory cortex but also premotor and motor regions, even in the absence of overt movement. Conversely, performing a musical sequence engages auditory regions even when no external sound feedback is present. This functional overlap suggests the existence of shared representations between perception and action.
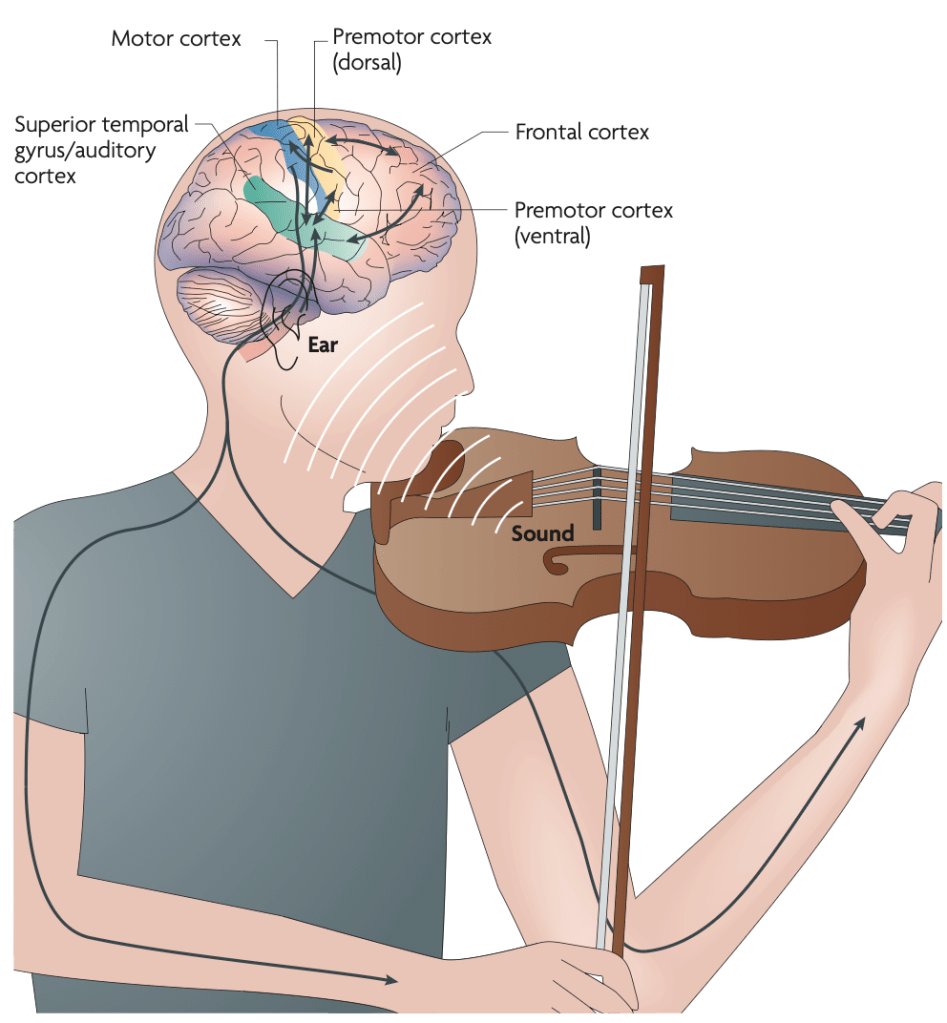
Image credit: Zatorre RJ, Chen JL, Penhune VB. When the brain plays music: auditory–motor interactions in music perception and production. Nat Rev Neurosci. 2007;8(7):547-558. doi:10.1038/nrn2152
When a violinist plays, for example, a highly integrated auditory–motor circuit is set into motion. The primary motor cortex executes the fine movements of the hand and fingers; the premotor cortex organizes the sequences required to produce each note; and prefrontal regions participate in the broader structural planning of the piece, sustaining context and interpretative intention. The cerebellum refines temporal precision and corrects errors within milliseconds, while the basal ganglia contribute to fluency, automatization, and the fine-tuning of the motor pattern.
In parallel, the auditory component does not merely register the produced sound. Beyond the primary auditory cortex, secondary auditory areas such as the planum temporale and regions of the superior temporal lobe are engaged in the fine analysis of pitch and melodic contour. Bilateral frontotemporal networks evaluate harmonic coherence, comparing what is sounding with what ought to sound. And the structures involved in rhythmic processing not only sustain the present movement but anticipate the next gesture before it occurs.
Thus, playing a musical instrument is not merely a matter of coordinating manual movements and listening to the result. It involves maintaining an internal representation of the expected sound, executing the corresponding action, and correcting it in real time. The musician’s brain does not simply respond to music: it simulates it, predicts it, and continuously adjusts it.
Yet there is an even more fascinating dimension of music—one in which a piece can sound in the absence of sound.
Auditory imagery⁸—the capacity to “hear in the mind”—is not merely a poetic metaphor, as we saw in The Pianist, when Szpilman mentally performs Chopin’s Andante spianato et Grande polonaise brillante, Op. 22 without touching an instrument. Auditory imagery is a well-documented neurobiological phenomenon. Functional magnetic resonance imaging studies have shown that imagining a melody activates broad regions that also participate when we actually hear it.
In particular, this overlap between musical perception and imagination is observed in secondary auditory areas of the temporal lobe. Moreover, internally generated music recruits a frontoparietal network that includes the dorsolateral prefrontal cortex, the supplementary motor area, and superior parietal regions. This network appears to exert “top-down” control, modulating activity within auditory areas. In other words, the prefrontal cortex does not merely retrieve a melody from memory—it sustains it, monitors it, and regulates it in real time.
Thus, it becomes clear that the brain does not merely recall a melody passively; it actively reconstructs it through functional connectivity between the right anterior superior temporal gyrus and prefrontal regions. This integrated frontotemporal circuit helps explain why Beethoven, completely deaf, was able to conceive the Ninth Symphony: music does not depend exclusively on the peripheral ear, but is constructed through complex cortico-subcortical networks capable of generating internal auditory representations.
Musical memory, therefore, is not solely episodic or semantic. It includes a vivid representational component that can activate auditory pathways in the absence of external stimulation, much as we can imagine a conversation or mentally rehearse a sentence. In the case of music, however, this internal representation may preserve pitch, tempo, and even timbre with extraordinary fidelity.
Likewise, when a patient with advanced Alzheimer’s disease recognizes a melody or manages to hum a song learned decades earlier, what we witness is not merely a sublime scene, but the activation of partially preserved networks—distinct from those that sustain recent episodic memory. Musical semantic memory relies on distributed networks within the neocortex, while the emotional dimension of the recollection engages limbic circuits that transport the individual into a timeless realm, where profoundly human feelings can be re-experienced—transcending time and, paradoxically, memory itself.
H.M.’s case taught us that memory extends beyond explicit recollection. The remarkable precision with which he was able to acquire new procedural memories despite the complete absence of both hippocampi also helps explain why Clive Wearing—and my patient—can continue to play their instruments despite severe amnesia. This occurs because music operates through the finely tuned interplay of multiple systems, capable of compensating for the loss of one.
Perhaps that is why my flutist patient can improvise a “Happy Birthday to You.” even though she cannot recall what occurred minutes earlier. Improvisation demands working memory, tonal anticipation, sensorimotor integration, and fine error correction. It requires, in other words, a brain still capable of dynamically sustaining music—even when autobiographical narrative has become fragmented.
Musical procedural memory, therefore, is not a recollection frozen in time, but a predictive process—a continuous dialogue between skills learned in the past, focused execution in the present, and melodic, harmonic, and tonal anticipation unfolding note by note. It is this finely orchestrated neurobiological conversation that becomes active when the body knows what to do before the mind can narrate it. Indeed, one might say that such a sequence can still unfold even when the episode that once gave rise to it has long since disappeared.
The explicit memory of rehearsal, of the teacher, of the first concert may be lost; the personal chronology of a life may erode. And yet, as the fingers assume the correct position, as breathing adjusts and the resolution of a phrase is anticipated, the brain organizes a coherent dynamic state in which perception, action, emotion, and prediction converge.
In that moment, it no longer matters that the hippocampus cannot consolidate new memories. It does not matter that episodic memory has grown fragile. Music is sustained by distributed networks that continue to dialogue with one another. Procedural memory is activated; working memory keeps the phrase alive; the cerebellum fine-tunes timing; the basal ganglia stabilize rhythm; the limbic system imbues the experience with emotional color. The person reappears.
Perhaps that is why, when my patient takes her flute, we are witnessing more than the preservation of a skill. We are observing the persistence of an identity. Music does not restore her memories, but it restores something equally essential: the internal coherence of being who she has always been.
And perhaps therein lies one of the most profound lessons of musical neuroscience: even when autobiographical memory fragments—when it becomes impossible to recall what day one is living in—time can be reorganized through music, offering something more valuable than a mere recollection: the capacity to reinhabit one’s identity and to be fully present.
References
- Alessandro L, Ricciardi M, Chaves H, Allegri RF. Acute amnestic syndromes. J Neurol Sci. 2020;413:116781. doi:10.1016/j.jns.2020.116781
- Chai WJ, Abd Hamid AI, Abdullah JM. Working memory from the psychological and neurosciences perspectives: a review. Front Psychol. 2018;9:401. doi:10.3389/fpsyg.2018.00401
- Budson AE, Price BH. Memory dysfunction. N Engl J Med. 2005;352(7):692–699.
- Squire LR. The legacy of patient H.M. for neuroscience. Neuron. 2009;61(1):6–9. doi:10.1016/j.neuron.2008.12.023
- Zatorre RJ, Salimpoor VN. From perception to pleasure: music and its neural substrates. Proc Natl Acad Sci U S A.2013;110(Suppl 2):10430–10437. doi:10.1073/pnas.1301228110
- Vuust P, Heggli OA, Friston KJ, Kringelbach ML. Music in the brain. Nat Rev Neurosci. 2022;23(5):287–305. doi:10.1038/s41583-022-00578-5
- Zatorre RJ, Chen JL, Penhune VB. When the brain plays music: auditory–motor interactions in music perception and production. Nat Rev Neurosci. 2007;8(7):547–558. doi:10.1038/nrn2152
- Herholz SC, Halpern AR, Zatorre RJ. Neuronal correlates of perception, imagery, and memory for familiar tunes. J Cogn Neurosci. 2012;24(6):1382–1397. doi:10.1162/jocn_a_00216
- Jacobsen JH, Stelzer J, Fritz TH, Chételat G, La Joie R, Turner R. Why musical memory can be preserved in advanced Alzheimer’s disease. Brain. 2015;138(8):2438–2450. doi:10.1093/brain/awv135
- Clark CN, Warren JD. Music, memory and mechanisms in Alzheimer’s disease. Brain. 2015;138(8):2122–2125. doi:10.1093/brain/awv164
- Groussard M, Chan TG, Coppalle R, Platel H. Preservation of musical memory throughout the progression of Alzheimer’s disease? Toward a reconciliation of theoretical, clinical, and neuroimaging evidence. J Alzheimers Dis.2019;68(3):857–883. doi:10.3233/JAD-180474
- Keith CM, McCuddy WT, Lindberg K, et al. Procedural learning and retention relative to explicit learning and retention in mild cognitive impairment and Alzheimer’s disease using a modification of the trail making test. Aging Neuropsychol Cogn. 2023;30(5):669–686. doi:10.1080/13825585.2022.2077297
- Harding EE, Kim JC, Demos AP, et al. Musical neurodynamics. Nat Rev Neurosci. 2025. doi:10.1038/s41583-025-00915-4
- Stein A. Music and trauma in Polanski’s The Pianist (2002). Int J Psychoanal. 2004;85(3):755–765. doi:10.1516/5M8R-60PW-0JA8-VA6F
Narrative References
Sacks O. Musicophilia: Tales of Music and the Brain. Knopf; 2007.
Darwin C. The Descent of Man, and Selection in Relation to Sex. John Murray; 1871.
Darwin C. The Autobiography of Charles Darwin. John Murray; 1887.
Szpilman W. The Pianist: The Extraordinary Story of One Man’s Survival in Warsaw, 1939–1945. Picador; 1999.
Recommended Audiovisual Material
🎹 Chopin – Andante spianato, Op. 22
Rubinstein A. Andante spianato et Grande polonaise brillante, Op. 22: Andante spianato in G Major, Tranquillo.YouTube. Published [year if identifiable]. https://youtu.be/tpXM5ymbRR4. Accessed February 5, 2026.
🎼 Chopin – Ballade No. 1, Op. 23
Zimerman K. Ballade No. 1 in G Minor, Op. 23. YouTube. https://youtu.be/G_uLCAZVh5Y. Accessed February 5, 2026.
🎬 The Pianist (2002) – “I was a pianist”
Scene in which Szpilman is discovered by Captain Hosenfeld.
Polanski R, director. The Pianist (2002). Scene: “I was a pianist” – Szpilman discovered by Captain Wilm Hosenfeld [online film clip]. YouTube. https://youtu.be/kJp_8AaeWI8. Accessed February 5, 2026.
🧠 Clive Wearing – Documentary
Clive Wearing – The Man with No Short-Term Memory. YouTube. https://youtu.be/Vwigmktix2Y. Accessed February 5, 2026.
Leave a comment